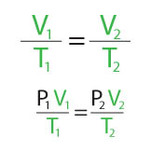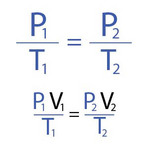First, Some Basics About Gases
|
A gas is a state of matter in which the particles are: 1) moving very fast (high kinetic energy), 2) very far apart (lots of space between them) and 3) not really attracted to each other (they have enough energy to overcome intermolecular forces that hold particles in liquids and solids closer together). They do not have a fixed shape or volume (instead they continue expanding to fill their container) and mix freely with other gases (no "oil and water don't mix" problems here!). They can also be easily compressed because there is so much space between their molecules.
|
Kinetic Molecular Theory (KMT)
|
The particles in a gas are always moving - no surprise there - and the study of kinetics helps us understand how those particles move and how their motion determines things like volume, temperature, and pressure.
Kinetic molecular theory is based on the following assumptions (..."but when we assume..." yes, I know, but sometimes we need to take a leap of faith and assume a few things). Seriously, though, models are imperfect but they make things easier to understand and certain behaviors easier to predict.
|
KMT and Gas Pressure
|
Gas pressure is the result of gas particles colliding with each other and with the walls of their container (Earth's atmosphere is one big "container" which is why we have atmospheric pressure).
These are the most common units of pressure, though there are others.
Standard Temperature and Pressure (STP)
|
KMT and Gas Laws
|
Dalton's Law of Partial Pressure
This is the easiest of the gas laws because we're only dealing with one variable - pressure. Dalton's law basically states that "the total pressure of a mixture of gases is equal to the sum of the partial pressures of the individual gases." (Pt = P1 + P2 + P3 + ...Pn). Calculations
|
|
The Combined Gas Law plus Boyle's, Charles' and Lussac's Laws
Next we're going to examine the relationship between PRESSURE (P), VOLUME V), and TEMPERATURE (T) of gas particles by studying the Combined Gas Law and it's spin-offs Boyle's Law, Charles' Law and Lussac's Law.
|
The Ideal Gas Law
|
|
Okay, much like the Easter Bunny, the Tooth Fairy and true Democracy, Ideal Gases do not exist. They just don't. So why the pretty story? Because the math needed to deal with real gases is horrifying! There are too many of real gases for one thing, and they have different intermolecular forces, and they come in a mathematically maddening variety of sizes and shapes, so to make our lives easier we idealize them. Trust me, it's okay to pretend.
Ideal Gases are defined as those that follow the Kinetic Molecular theory and Boyle's and Charles's Laws exactly. Their behavior can be quantified using the equation PV=nRT where P= Pressure, V= Volume (in Liters), n= Moles, R=Gas Constant, and T=Temperature (in Kelvins). Values for "R" vary with the Pressure unit you are using. Notice the only thing that changes in the R unit is the unit for pressure.
Calculations: Sample problem: 5.00 g of neon is at 256 mm Hg and at a temperature of 35º C. What is the volume? Important! Always convert: a) grams to moles (divide grams by the molar mass), b) º C to Kelvin (add 273.15), and c) mL to L. Step 1: Write down your given information and convert as needed:
|
Advanced Applications - Challenge your brain!
|
|
Gas Stoichiometry
Remember how we used stoichiometry to quantify and predict amount of reactants and products using balanced chemical equations? You can perform the same types of calculations can be used in reactions involving gases.
|